Nobel Prize 2021 for chemistry goes to scientists Benjamin List and David MacMillan for developing asymmetric organocatalysis

Nobel laureates Benjamin List and David MacMillan have discovered a third type of catalyst that will help making chemical reactions eco-friendly
The committee has announced the Nobel Prize 2021 for Chemistry to American scientists Benjamin List and David WC Macmillan for developing asymmetric organocatalysis today.
These organic catalysts would make chemistry greener and impact the pharmaceutical research kingdom. Independently of each other, Nobel laureates List and MacMillan have developed a third type of catalysis – asymmetric organocatalysis – a precise new tool for molecular construction.
What are organ catalysts?
Until recently, researchers have believed that there are only two kinds of catalysis – metal and enzymes. But in 2021, the scientist duo, independently of each other, has developed the third one. An organic catalyst has a stable structure of carbon atoms. The stable structure of carbon atoms aided in attaching more active chemical groups that commonly have elements like oxygen, nitrogen, sulphur, or phosphorus. The organ catalysts are both eco-friendly and cheap to produce.
“The rapid expansion in the use of organic catalysts is mainly due to their ability to conduct asymmetric catalysis. When molecules are built, situations often occur where two different molecules can form, which – just like our hands – are each other’s mirror image,” the press release read. Often the chemist will require only one molecule to produce pharmaceutical products.
Both Benjamin List and David MacMillan have shown that organic catalysts can lead a horde of chemical reactions to help researchers constructing anything from new pharmaceuticals molecules to light-capturing molecules in solar cells more efficiently.
Benjamin List experimented with amino acid
Benjamin List worked with catalytic antibodies, large molecules made by hundreds of amino acids. Usually, now, many enzymes catalyse chemical reactions are driven by a single or a few individual amino acids. In the early 1970s, an amino acid called proline was used as a catalyst. List tested the catalysing capacity of proline in an aldol reaction. The aldol reaction has carbon atoms from two different molecules bonded together.
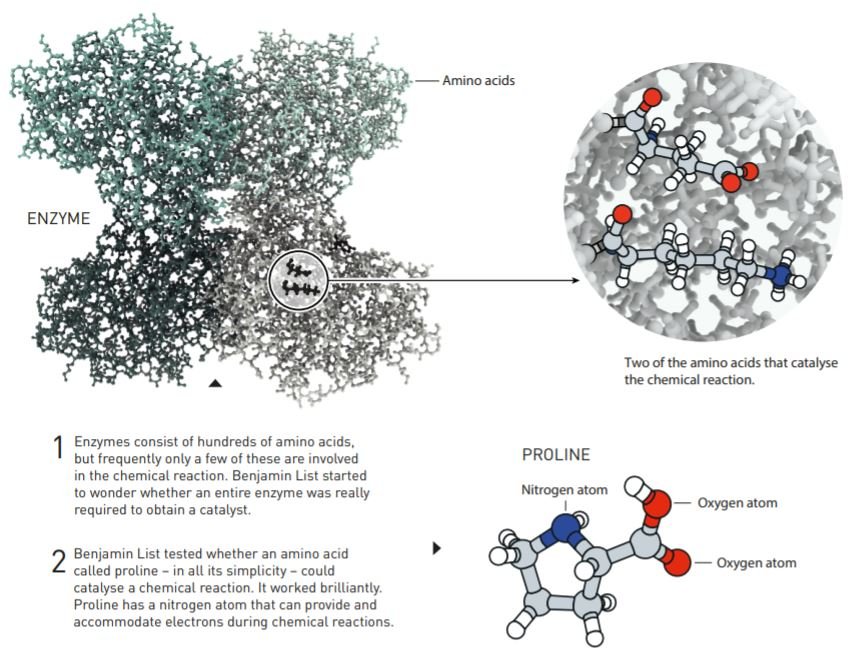
The experiment worked straight away demonstrating amino acid’s ability to drive asymmetric catalysis. With his experiments, Benjamin List discovered proline as an efficient catalyst and its ability to drive asymmetric catalysis reactions. “The design and screening of these catalysts are one of our future aims,” List said while publishing his results.
David MacMillan coined the term ‘Organocatalysis’
David wanted to build a simple catalyst after seeing how metal catalysts were left behind in industrial surroundings due to feasibility issues. Therefore, he developed simple organic molecules which could temporarily provide or accommodate electrons, just like metals.
These organic molecules have simple elements but can demonstrate complex properties depending on the bonding of the property elements. But they should be able to form an iminium ion. Iminium is an important functional group for the synthesis of heterocyclic ring systems, according to ScienceDirect. Thus David decided to select molecules with nitrogen atoms. These nitrogen atoms have a high affinity for electrons.
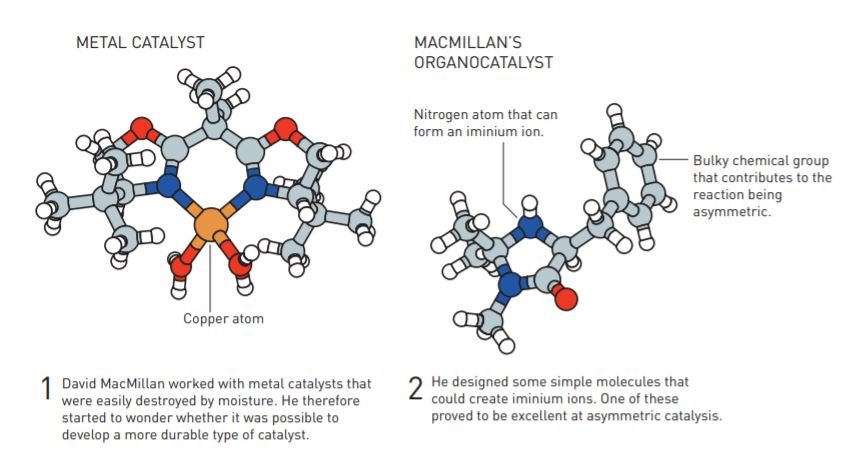
He then tested the ability of organic molecules with various properties to a Diels–Alder reaction. As hoped, some of them were excellent at asymmetric catalysis. When creating two possible mirror images, one of them had a composition similar to more than 90% of the parent product. He coined the term ‘organocatalysis’ while publishing the result.
Feature Image: Benjamin List And David MacMillan (Source: Twitter/NobelPrize)

